by J.G. Davis, R.M. Waskom, and T.A. Bauder* (5/12)
Quick Facts…
- Sodic soils are poorly drained and tend to crust.
- Sodic soils respond to continued use of good irrigation water, good irrigation methods, and good cropping practices.
- Sodic soils are often reclaimed by adding a calcium-based soil amendment.
Soils with high levels of exchangeable sodium (Na) and low levels of total salts are called sodic soils. Sodic soils may impact plant growth by: 1.) Specific toxicity to sodium sensitive plants; 2.) Nutrient deficiencies or imbalances; 3.) High pH; and 4.) Dispersion of soil particles that causes poor physical condition of the soil.
Sodic soils tend to develop poor structure and drainage over time because sodium ions on clay particles cause the soil particles to deflocculate, or disperse. Sodic soils are hard and cloddy when dry and tend to crust. Water intake is usually poor with sodic soils, especially those high in silt and clay. Poor plant growth and germination are also common. The soil’s pH is usually high, often above 9.0, and plant nutritional imbalances may occur. A soil pH above 8.4 typically indicates that a sodium problem exists. The term “alkali” is often used to describe soils that are high in salt but sometimes people use the term to mean high pH and at other times to mean high sodium. “Black alkali” refers to a sodic soil condition where organic matter has spread and is present as a dusty material on the soil surface.
Sodium levels in soil are often reported as the sodium adsorption ratio (SAR). This is a ratio of the amount of cationic (positive) charge contributed to a soil by sodium, to that contributed by calcium (Ca) and magnesium (Mg). The SAR is determined from a water extract of a saturated soil paste. If the SAR is above 13, the soil is classified as sodic (Table 1). However, sodium can cause soil structure deterioration and water infiltration problems. at SAR levels below 13 in some cases. The severity of symptoms with high SAR soils depends upon many sitespecific factors including soil type, texture, drainage conditions and irrigation water quality. Some labs report high sodium levels as ESP (exchangeable sodium percentage). An ESP of more than 15 percent is sometimes used to classify a soil as sodic. This means that sodium occupies more than 15 percent of the soil’s cation exchange capacity (CEC). Be aware that sensitive plants may show injury or poor growth at even lower levels of sodium.
Sodium Hazard
Analyzing the soil for both soluble salts and sodium levels helps identify the specific soil problem and its severity. To find out if a problem exists, take a composite sample of several cores, 6 to 8 inches deep, from the affected area. In many cases, comparing soil samples from the affected area to surrounding normal appearance areas is beneficial in diagnosing the problem. Other information including soil texture, cation exchange capacity, type of clays present, calcium carbonate content, organic matter, depth to ground water, and soil profile information will help determine a recovery program. Some of this information may be obtained in the county soil survey available through your local USDA-Natural Resources Conservation Service (NRCS) office.
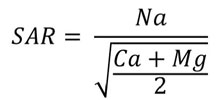
Figure 1. Formula for calculating SAR (sodium adsorption ration). |
The sodium hazard of soil usually is expressed as the sodium adsorption ration (SAR). This is the proportion of water soluble Na to Ca plus Mg in the soil. The formula used to calculate SAR is shown in Figure 1.
Ions in the equation are expressed in millequivalents per liter (meq/L) and are obtained from a saturated paste soil extract. To convert ppm or mg/L Na+ to meq/L, divide by 23; for Ca++ divide by 20; and for Mg++ divide by 12.2.
Specific Ion Effects
Sometimes a specific ion can have toxic reactions in certain plants. Sodic soils can cause specific ion toxicity in sensitive crops such as potatoes, beans and woody plants such as vines and stone fruits. High sodium levels compete with calcium, magnesium, and potassium for uptake by plant roots. Therefore, excess sodium can prompt deficiencies of other cations (positively charged nutrients). High levels of other cations (calcium, magnesium, potassium) can also cause imbalances and induce nutrient deficiencies.
| Table 1. General classification for sodium hazard of soil based on SAR values. | ||||
| Classification | Sodium adsorption ratio (SAR)2 | Electrical conductivity (dS/m)1 |
Soil pH | Soil physical condition |
| Sodic | ≥13 | <4.0 | >8.5 | poor |
| Saline-Sodic | ≥13 | >4.0 | <8.5 | varies |
| Slightly Saline | <13 | 2 – 4 | <8.5 | normal |
| Saline | <13 | >4.0 | <8.5 | normal |
| High pH | <13 | <4.0 | >7.8 | varies |
| 1dS/m = mmho/cm | ||||
| 2If reported as Exchangeable Sodium Percentage or ESP, use 15% as threshold value. | ||||
Managing Sodic Soils
There are usually three options for correcting soil-related problems:
- change the plant species to a more tolerant species, or,
- change the variety to a more tolerant variety or,
- change the soil.
Often, changing the soil is the most difficult of these options.
When soils are high in sodium, the goal is to replace the sodium with calcium and then leach the sodium out. There are two possible approaches for doing this:
- dissolve the limestone (calcium carbonate) or gypsum (calcium sulfate) already present in the soil or,
- add calcium to the soil.
If free lime is present in the soil, it can be dissolved by applying sulfur or sulfuric acid. Sulfur products reduce the pH which dissolves the lime, thus freeing up the calcium. If free lime or gypsum is not present in adequate amounts as determined by a soil test, then add calcium.
The most common form of calcium used for this purpose is gypsum. Although calcium chloride, which reacts more quickly, can also be used it is usually more expensive. After broadcasting the calcium source on the soil surface, mix it, and make sure adequate moisture is present to dissolve it.
Recovering a foot depth of sodic soil on one acre requires approximately 1.7 tons of pure gypsum (CaSO4-2h2O) for each milliequivalent of exchangeable sodium present per 100 grams of soil.
Example gypsum requirement calculation:Your soil has a CEC of 18 milliequivalents per 100 grams and SAR of 26, and you desire an SAR of approximately 10 following treatment. (In these calculations it is correct to assume SAR is roughly equivalent to ESP.) ESP of 26% – desired ESP of 10% = ESP of 16, or 16% exchangeable Na must be replaced with calcium (Ca) to achieve the desired SAR. 0.16 (16%) x 18 meq CEC /100g = 2.88 meq Na/100 g soil that must be replaced. *1.7 tons CaSO4 x 2.88 meq Na = 4.9 tons of gypsum. Thus, about 5 tons of pure gypsum per acre would be required to reclaim the top 12 inches of this soil. Be sure to adjust this calculation for lower grades of gypsum and different soil depths. *As a general rule of thumb, 1.7 tons of gypsum is required per meq of sodium. |
Once the gypsum is applied and mixed, sufficient quality water must be added to leach the displaced sodium beyond the root zone. Restoration of sodic soils is slow because soil structure, once destroyed, is slow to improve. Growing a salt-tolerant crop in the early stages of reclamation and cultivating in crop residues or manure adds organic matter which will increase water infiltration and permeability to speed up the reclamation process.
Make sure drainage is adequate prior to amending the soil, and after application of a sulfur product or a calcium source, leach the sodium out with good, quality water. Success in reclaiming non-irrigated sodic or saline-sodic soils with gypsum application may be possible on coarse textured soils that receive precipitation in excess of soil water holding capacity.
Remember:
- Adding sulfur products only makes sense when:
- a) a soil is sodic and has free lime present or, when
- b) a soil is basic (high pH).
- Adding calcium sources, such as gypsum or calcium chloride to saline (not sodic) soils only increases the salt content further and aggravates the salinity problem.
In many cases, the common practice is to apply sufficient amendment to remove most of the adsorbed sodium from the top 6 to 12 inches of soil. This improves the physical condition of the surface soil in a short period of time and permits the growing of crops. Continued use of quality irrigation water, good irrigation methods, and cropping practices further displaces adsorbed sodium. In some cases, it may be necessary to restore the soil to greater depths to obtain adequate drainage and root penetration.
Incorporating crop residues or plowing under manure, compost, green manure or cover crops may improve the tilth and increase water infiltration of sodium affected soils, especially when combined with other reclamation practices. It is generally better to grow a sodium tolerant crop during reclamation than to leave the field fallow. Deep plowing to disrupt restrictive claypans and to mix calcium from deeper soil layers has also been used effectively in some situations.
| Table 2. Amount of amendments required to supply one pound of soluble calcium. | ||
| Amendment | Purity* % | Pounds |
| Gypsum | 100% | 4.3 |
| Calcium chloride | 100% | 3.7 |
| Sulfur | 100% | 0.8 |
| Sulfuric acid | 95% | 2.6 |
| Lime sulfur | 24% sulfur | 3.3 |
| *If the amendment has a purity different from that indicated on the table, determine the amount needed to supply one pound of soluble calcium by dividing the percent purity in the table by the percent purity of the material to be applied and multiply this by the number of pounds shown in the table. |
||
Types of Amendments
Several commercial products are now on the market for amending sodic and saline-sodic soils. The only function of scientifically proven amendments is to provide soluble calcium to replace exchangeable sodium adsorbed on clay surfaces. There are two main types of amendments: those that add calcium directly to the soil and those that dissolve calcium from calcium carbonate (CaCO3) already present in the soil.
Calcium amendments include gypsum (hydrated calcium sulfate) and calcium chloride. Gypsum is moderately soluble in water. Calcium chloride is highly water soluble and fast acting, but it generally is too expensive for most situations.
Acid-forming, or acidic amendments, include sulfuric acid, elemental sulfur, and calcium carbonate-sulfur. Sulfuric acid reacts immediately with the soil calcium carbonate to release soluble calcium for exchange with sodium. Elemental sulfur must be oxidized by soil bacteria and react with water to form sulfuric acid. The formation of sizeable amounts of sulfuric acid from elemental sulfur may take several months to several years.
Calcium carbonate-sulfur must go through essentially the same process as elemental sulfur and also is considered a slow-acting amendment. Calcium carbonate must be present in the soil when acid or acid-forming amendments are added.
Choose the amendment mainly on the basis of the cost of the soluble calcium furnished directly or indirectly by the amendment and the speed of the reaction. Also consider ease of application.
*J.G. Davis, Colorado State University Extension soils specialist and professor, soil and crop sciences; R.M. Waskom, Colorado Water Institute director; and T.A. Bauder, water quality specialist.7/03. Revised 5/12.
Colorado State University, U.S. Department of Agriculture, and Colorado counties cooperating. CSU Extension programs are available to all without discrimination. No endorsement of products mentioned is intended nor is criticism implied of products not mentioned.
Go to top of this page.





