by Chakradhar Mattupalli1 and Amy O. Charkowski2 (4/19)
Quick Facts…
- Sanitation and exclusion are key to prevent introduction of pest inoculum in greenhouse potato minituber production.
- Potable ground water should be used to irrigate greenhouse grown potatoes to reduce risk caused by bacterial and fungal pathogens.
- Use of virus-free tissue culture plantlets and timely management of aphids and thrips will reduce the chance of virus infestations.
- Greenhouses should be maintained between 22-28°C, which promote potato plant development while limiting diseases caused by Pythium and Spongospora.
- Avoid growing plants such as ornamentals while growing potatoes in the greenhouse as they can host destructive bacterial and viral pathogens.

Figure 1: Potato plants growing in the greenhouse. Inset image shows potato minitubers.
One of the first steps of seed potato production in North America relies on production of minitubers in greenhouses (Fig. 1). Micro-propagated plantlets are planted into potting mix or hydroponic systems to produce minitubers that should be free of all important potato diseases. Some simple management practices can aid greenhouse growers in producing a healthy minituber crop.
Disease management
There are only a few potato pathogens that are common in greenhouse-grown potatoes. Sanitation and exclusion can aid in management of most of these pathogens. The most common source of these pathogens is the outside environment surrounding the greenhouse or unsanitized surfaces inside the greenhouse. Pathogen management tends to be easier in crops grown between January and May, when pest populations outside are low.
Potato viruses and viroids
Multiple potato viruses and a potato viroid can occur in greenhouses and most of them can easily spread between plants on worker hands or by insect vectors. Growers should maintain a zero tolerance for potato viruses in greenhouses.
- Potato virus S (PVS), Potato virus X (PVX), and Potato spindle tuber viroid (PSTVd) all spread easily in greenhouses when workers touch plants or when the plants touch each other. PVS and PVX rarely cause foliar symptoms. Although these viruses do not cause much damage on their own, they may be synergistic with other pathogens. For example, if both PVX and Potato virus Y (PVY) are in a plant, the symptoms will be much more severe than in plants infected with only one virus. If PVS, PVX, or PSTVd are present, the likely source is the initial tissue culture plantlets or seedlings used to plant the greenhouse crop.
- PVY and Tomato spotted wilt virus (TSWV) are sometimes found in greenhouses (Fig. 2A). Generally, these viruses enter greenhouses due to aphid (PVY) or thrip (TSWV) infestations. Good insect management will reduce the chance that these viruses will be present in a greenhouse crop.
- Greenhouse workers can reduce the chance of virus spread in greenhouses by washing their hands or changing gloves between potato lots or greenhouses and by sanitizing pots, benches, and floors between crops. A plant-free period also aids in virus management since it will aid in elimination of aphid vectors.
Bacterial pathogens

Figure 2: Some diseases affecting potato minituber production. A) Mosaic on potato leaves caused by PVY (Photo Courtesy: Ana C. Fulladolsa) B) Potato affected with bacterial soft rot C) Potato tuber showing powdery scab lesions.
The bacterial soft rot and blackleg pathogens in the Pectobacterium and Dickeya genera can be found in greenhouses (Fig. 2B). Both tend to cause symptoms under greenhouse conditions, but it is also possible that infected plants will remain symptomless. The likely source for these pathogens is irrigation water. Whenever possible, potable ground water should be used to irrigate greenhouse-grown potatoes to reduce the chance of bacterial and fungal pathogens. Greenhouse-grown plants should not be irrigated with surface water. If possible, greenhouse irrigation water should be purified by a reverse osmosis system. Exclusion of Clavibacter michiganensis subsp. sepedonicus (causal agent of bacterial ring rot of potato) is of extreme importance. The pathogen can be introduced into a greenhouse through contaminated shoes, clothing or equipment. Hence, proper exclusion and disinfection protocols must be followed to prevent contamination.
Fungal pathogens
Fungal pathogens sometimes attack potato foliage in greenhouses. The most common foliar fungal disease is powdery mildew. This disease is typically mild on potato and can be managed by spraying the leaves with parafinnic oil. Early blight and gray mold can also be common on greenhouse grown potato, which may be managed with Pyrimethanil fungicides. Colletotrichum coccodes, which causes black dot of potato, can be present in potting mix and may cause mild black dot symptoms on potato. The black dot pathogen can also be present in hydroponic systems, where it will inhibit root development and plant growth and can severely limit tuber production.
Pythium, Spongospora and PMTV in potting mix
Pythium is present in peat and sphagnum moss and can cause damping off of potato transplants or cuttings. The most severe Pythium problems occur if potting mix and hydroponic systems are used in the same greenhouse operation since Pythium causes severe losses if it contaminates hydroponic systems. Spongospora subterranea, the cause of powdery scab, is commonly present at low levels in peat-based potting mix (Fig. 2C). It can cause severe disease if the greenhouse is maintained at 18°C or below. Warmer temperatures will inhibit this pathogen. Spongospora can multiply to high levels under ideal greenhouse conditions, going from 1 sporosori per gram of potting mix to over 200,000 sporosori per gram in about 3 months. Therefore, potting mix should not be reused or spread onto agricultural fields. S. subterranea is the vector of Potato mop-top virus (PMTV), which causes tuber necrosis. It is not known yet whether potting mix can also carry PMTV.
Insect Management
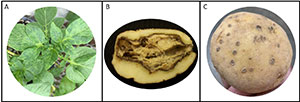
Figure 3: Common insect pests in potato greenhouse production. A) Green peach aphid B) Thrips C) Spider mite (Photo courtesy: www.nwpotatoresearch.com/insects/; A. Jensen).
Two insect pest groups are common in greenhouses, aphids and thrips (Fig. 3A and 3B). Both insects can cause damage through feeding and can spread viruses. Insecticides, with active ingredients such as azadirachtin, clothianidin, lambda-cyhalothrin, dimethoate, flonicamid, malathion, phorate, imidacloprid, tolfenpyrad etc, are available and can be used to protect greenhouse-grown potatoes against aphids and thrips. Always read the product label prior to using any pesticide. In addition to chemical control, several predators, parasitoids, entomopathogenic fungi may be employed to manage these pest populations. Some growers allow a plant free period of at least a month between subsequent plantings to eliminate pest infestations.
Aphids spread many viruses, including PVY and Potato leafroll virus (PLRV). If aphids are found in a greenhouse, the infested plants should be discarded and the remaining plants should be treated with an insecticide. If the infestation is severe, the entire crop should be destroyed. If aphids are found, the remaining plants should be tested for PVY and PLRV. It takes at least three weeks after aphid transmission to be able to detect virus spread, so virus testing should either happen one month after the infestation is noted or the tubers should be tested. If virus is detected, the minitubers should not be planted the following season. Aphids do not survive well in greenhouses without living plants and a plant free period of at least a month may help to eliminate pest infestations. Aphids are attracted to yellow and placement of yellow sticky cards in a greenhouse will aid in early detection of aphids.
Thrips damage plant leaves through feeding and also spread TSWV. This virus can cause necrotic flecks or rings on potato leaves and necrotic spots or rings inside potato tubers. Thrips can survive for long periods of time without green plants. If a greenhouse infestation occurs, be sure to remove all plant material from the greenhouse and thoroughly sanitize the greenhouse before planting new potato plantlets. Thrips are attracted to blue and placement of blue sticky cards in a greenhouse will aid in early detection of thrips.
A third common greenhouse pest is the spider mite (Fig. 3C). This arthropod feeds on growing plants and toxins from its feeding will distort or kill the plants. Symptoms of spider mite infestation include webbing, stippling and bronzing of leaves. Spider mites can be managed with pesticides containing the active ingredients spiromesifen, hexythiazox or package mixes containing bifenthrin and zeta-cypermethrin. In addition, oil sprays with neem oil, rosemary oil and peppermint oil may be used to suppress spider mite populations.
Greenhouse management
Pest exclusion
One of the golden rules of pest management is to prevent the introduction of pest inoculum. Ideally, greenhouses used for potato minituber production should be isolated from all field agriculture.
- Pathogens can enter greenhouses on or in seeds and tubers. For potato production in greenhouses, use only micro-propagated plantlets coming from mother plants that have been tested and found to be free of important potato pathogens.
- Insects and pathogens can enter greenhouses on greenhouse workers. To prevent this, workers should complete greenhouse work before starting field work or the workers should not enter greenhouse if they have been working in the field/warehouse unless proper sanitation of hair/clothes/shoes etc. has occurred. This will help workers avoid carrying in aphid, thrips, or other insects from field-grown plants into the greenhouses on their clothes or hair.
- Greenhouses should have double door entries that cannot be opened at the same time to help block the entry of pests into the greenhouse. Lab coats, boots, hairnets, and gloves can be kept in the entry between door 1 (outside to entryway) and door 2 (entryway to greenhouse) to provide workers with clothing that will reduce the chance that insects or pathogens will spread from their clothes or shoes to the greenhouse plants. Lab coats can be treated with permethrin clothing spray to reduce the spread of insects into and among greenhouses on greenhouse workers.
- Greenhouses should be maintained with positive pressure. This will stop insects and pathogens from being transported by air into the greenhouse. Insect-proof screen should be used over fan vents to reduce the chance that thrips or aphids can enter the greenhouse. The greenhouse walls and roof should not be cracked or have holes. All gaps between wall and roof, or at entry points of plumbing and wiring should be sealed. Growers should clear all weeds from the perimeter of the greenhouse and not use ornamental plants to landscape near their greenhouse buildings.
- Different times of year affect the risk of pest introduction into greenhouses. Pest populations are highest in late summer and early fall in seed potato producing regions in North America. In contrast, pest numbers are very low in later winter or early spring. Greenhouse crops are easiest to manage if they are planted in late January or early February and harvested by May. Crops planted anytime between May and September will face a higher risk of pest introduction.
- Growers who produce seed potatoes, should not grow other crops in their greenhouse while growing potato. Other plant species commonly grown in greenhouses can host destructive pathogens that are not yet present in the North American potato industry. For example, geraniums and other ornamentals can carry Ralstonia solanacearum, a bacterial pathogen causing brown rot of potato. Ornamental plants can also carry Dickeya solani, an aggressive pathogen that causes potato blackleg. Viruses that cause tuber necrosis are common in ornamental plants too. For example, Tobacco rattle virus (TRV) and Tomato spotted wilt virus (TSWV) both commonly infect ornamental plants.
Sanitation
Greenhouses should be designed for ease of sanitation between crops. Ideally, floors should be concrete and there should be as little wood present as possible. If gravel floors are used, they should be covered in plastic since spores of pathogens such as Sclerotinia or insects, such as thrips, can survive between crops in gravel floors. Floors, irrigation equipment, pots, and any other equipment used for crop production should be sanitized between crops. Sanitizers that include chlorine bleach, quaternary ammonium compounds, hydrogen dioxide may be used for sanitizing greenhouse surfaces and benches. Follow the label directions when using sanitizers and never mix different types of sanitizers since this can result in toxic fumes. Sanitizers are typically developed for use in hospitals or food preparation facilities. They are rarely tested for efficacy against potato pathogens. However, some potato pathogens are closely enough related to human pathogens that the test information is still useful. For example, the bacterial blackleg pathogens may have similar sensitivity to SaniDate®5.0 as Escherichia coli.
Greenhouse environment management
Greenhouse conditions can suppress or favor disease development or can cause physiological symptoms in potato plants. Greenhouses should be maintained between 22-28°C, a range that suppresses disease caused by Pythium and Spongospora, but that is not too hot for potato plant development. High temperatures and high humidity can cause leaf edema or aerial sprouts on potato plants. Typically, neither condition will cause a significant yield reduction, but these physiological disorders are indications that greenhouse temperature and/or humidity management should be improved.
Potato yields in greenhouses are very sensitive to fertilization levels. For example, nitrogen levels have a dramatic impact on tuber yields in greenhouses. Too much nitrogen will result in excessive foliage production and reduced tuber yields. The ideal nitrogen level differs among varieties with either potting-mix based systems or hydroponic systems. Potatoes are also sensitive to day length and may not tuberize efficiently, especially in hydroponic systems, if days are longer than 12 hours.
Genetic mutations in potato
Potatoes are typically multiplied with micropropagation (tissue culture) prior to planting in a greenhouse. Genetic mutations sometimes occur in the initial planting stock (the mother plant) prior to multiplication. Mutations that cause obvious yield or quality losses occur at a rate of about 0.2% of clones per year. The physiological effect of mutations are generally not possible to detect until the plants are grown in the greenhouse or field. Growers should keep different clones of the same variety separate in greenhouses. Clones that have unexpected physiology, including tuber or leaf shape, flower color, or yield should be discarded. Growers should revive their clones from healthy tubers of ideal color, shape, and plant morphology regularly (typically every five years) to reduce the accumulation of genetic mutations in micropropagated plants.
Minituber storage
Minitubers harvested from hydroponic systems tend to store better if they are exposed to light for a few days and allowed to turn green. This can be done inside the greenhouse, if space is available, or in any clean indoor space. The tubers can be stored in bins, mesh bags, or even plastic bags with holes to provide airflow. Greenhouse-grown minitubers can be stored under similar conditions as field-grown seed potatoes. However, they should be stored separately from field-grown tubers if possible because inoculum of pathogens such as Helminthosporium solani, the causal agent of silver scurf, can spread between storage bins by warehouse air systems.
1Chakradhar Mattupalli, Research Professor and Extension Specialist – Plant Pathology, San Luis Valley Research Center and 2Amy Charkowski, Professor and Department Head, Bioagricultural Sciences and Pest Management. (4/19).





